The OSHA Vaccine or Test ETS was blocked by the Supreme Court (SCOTUS) last week. SCOTUS essentially reapplied the temporary injunction that blocks OSHA from enforcing the ETS. While it may not be gone for good, many think it unlikely the ETS will survive. Several have supposed that it is more likely that OSHA will begin the process of adopting an infectious disease standard sometime in the near future.
While this is good news for general industry, the healthcare industry must also look at the Center for Medicare and Medicaid Services (CMS). On Friday, January 14, 2022, CMS issued a memorandum to the Interim Final Rule – Medicare and Medicaid Programs; Omnibus COVID-19 Health Care Staff Vaccination.
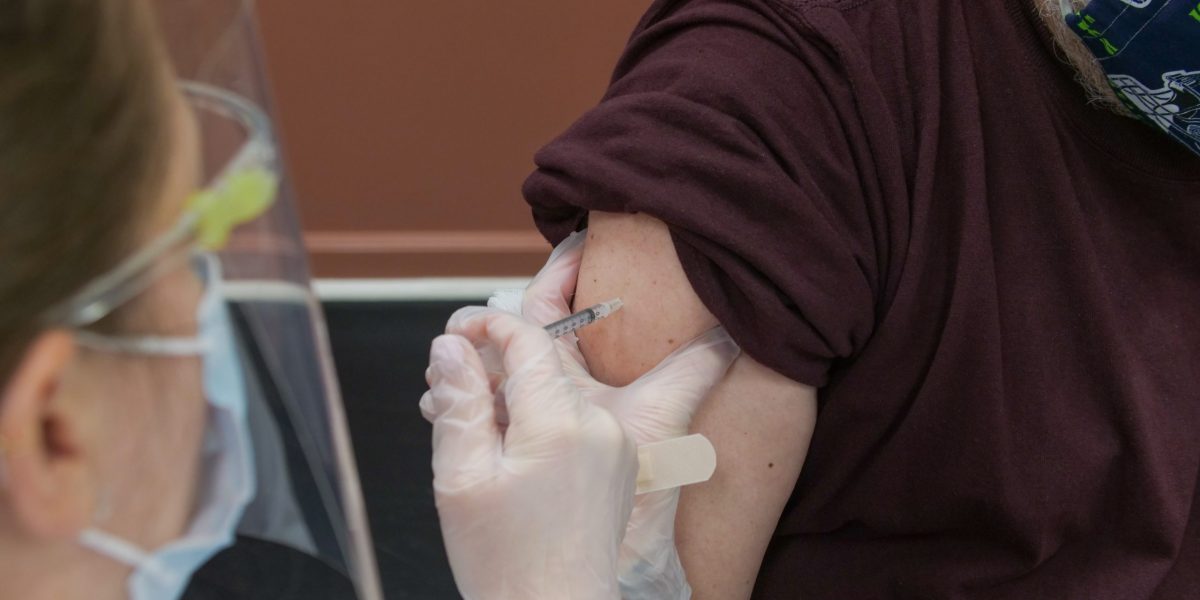
The memorandum states that within 30 days after issuance, or February 14, 2022, facilities must have implemented policies and procedures for ensuring all staff regardless of clinical responsibility or patient contact are vaccinated for COVID-19 and 100% of staff have received at least one dose of a COVID-19 vaccine, or have a pending/granted qualifying exemption, or identified as having a temporary delay as recommended by the CDC.
Within 60 days of issuance, or March 15, 2022, facilities must ensure 100% of staff have received the necessary doses to complete the vaccine series, or have a pending/granted qualifying exemption, or identified as having a temporary delay as recommended by the CDC. If less than 100% of staff have received at least one dose of a single-dose vaccine, or all doses of a multiple-dose vaccine series, or have a pending/granted qualifying exemption, or identified as having a temporary delay as recommended by the CDC, the facility is non-compliant under the rule. A facility that is above 90% and has a plan to achieve a 100% staff vaccination rate within 30 days would not be subject to additional enforcement action.
Within 90 days and thereafter following issuance of this memorandum, facilities failing to maintain compliance with the 100% standard may be subject to enforcement action.
Facilities must track and securely document any staff member who receives a booster dose as recommended by the CDC to include specific booster received as well as administration date.
DNV accredited hospitals must be in compliance with these requirements and DNV will begin surveying to the requirements on February 14, 2022.
Joint Commission accredited hospitals must be in full compliance with these requirements by April 14, 2022, however, beginning January 27, 2022, for applicable deemed program surveys in progress on that day, The Joint Commission will begin surveying to the COVID–19 Health Care Staff Vaccination interim final rule published by CMS on November 5, 2021.